Oncology
We empower patients to self-manage their symptoms,
using evidence-based algorithms, to improve their quality of life,
treatment exposure and ultimately increase survival.
Symptom management, a key opportunity for better care
At Voluntis, we truly believe that giving the right support to patients can make a difference in their treatment journey. Experiencing side effects is very common when undergoing anti-cancer treatment, so our approach in oncology is based on self-management of symptoms to make patients more autonomous and active in their disease.
Our digital therapeutics in oncology embed scientifically-validated algorithms, built under our versatile and modular Theraxium platform, providing real-time, personalized recommendations to patients, so they know what to do and how to manage their symptoms, anytime and anywhere.
Treatment-related symptom management is a challenge faced by all stakeholders within healthcare systems that must respond to an ever-increasing demand. Promoting self-management helps address this challenge by facilitating communication between care teams and patients, encouraging shared decision-making and optimizing resource utilization by focusing on most critical cases.
of advanced practitioners report symptom management as the most performed task (1)
Up to 91% of patients experience cancer-related symptoms (2)
of unscheduled phone calls are caused by treatment-related symptoms (3)
- ACCC 2018 Trending Now in Cancer Care Survey
- Stark et al. “The Symptom Experience of Patients with Cancer.” J Hosp Palliat Nurs. 2012 Jan-Feb; 14(1): 61–70
- Remy C, Borniard J, Perez J. Analysis of Unscheduled Telephone Calls Received by a Specialized Cancer Pain Nurse. Pain Manag Nurs. 2020 Jun;21(3):255-258. doi: 10.1016/j.pmn.2019.07.009. Epub 2019 Aug 28. PMID: 31473170
An active approach to empower patients
Our active approach in oncology aims to enable patients to take actions in their disease and to better manage their side effects, thus improving treatment experience.
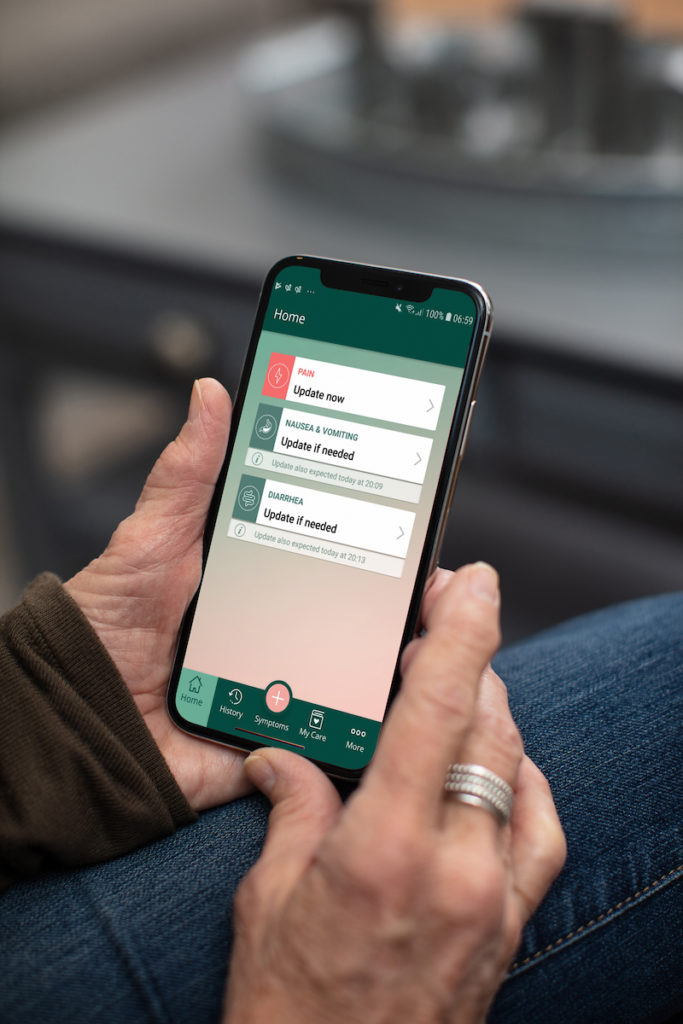
Our digital therapeutics are based on a mobile app for patients and a web portal for care team. Whether they are starting a new treatment or already under therapy, patients receive personalized and real-time recommendations to manage their issues and support remote monitoring by the care team. Leveraging embedded evidence-based clinical algorithms, the solution can help patients:
- Identify, qualify and report their symptoms
- Take action to self-manage their symptoms
- Know when they need to contact their care team
- Gain knowledge about their disease
Patients report symptom data in their smartphone app to receive real-time and actionable recommendations according to their profile and current condition. Care teams can focus the progress of their patient population from their dashboard thanks to their web app, without the need to download anything.
With the automatic feedback loop providing actionable guidance, our digital therapeutics have the objective of fostering patient engagement. The care team can focus on most critical cases as most interventions are delivered automatically. For each patient, the assessment of symptom severity is based on their individual profile and care plan, configured by the care team when creating the patient profile.
With their digital therapeutics in hands, patients also get access to additional features to better understand their disease and symptoms such as infocards and educational content from reliable sources.
The unique data generated by our digital therapeutics is collected and analyzed to identify trends for each patient and allows the adaptation of the remote monitoring performed by the care team. Our digital therapeutics aim at improving patients’ quality of life, treatment adherence and strengthening collaboration with healthcare teams while reducing care costs.
“It’s easy to use, very convenient, especially if you are not feeling well”
“Makes all life easier”
Oleena users
To learn more about our in-house digital therapeutic, Oleena®, please visit the dedicated website: www.oleena.com.
Oleena® is a prescription mobile app intended for use by healthcare professionals and their adult patients with cancer as an aid for the management of symptoms experienced during the oncology treatment phase. Before use, please carefully read product instructions available in the “More” tab of the app. Oleena is not to be used during pregnancy and by patient under 18 years old. Oleena is a registered trademark of Voluntis. For distribution in the US only.
A feasibility assessed approach for patients with ovarian cancer
In collaboration with AstraZeneca and the US National Cancer Institute (NCI), Voluntis developed eCO*, a DTx for women undergoing treatment for recurrent platinum-sensitive high-grade ovarian cancer in clinical trial of cediranib in combination with olaparib. eCO was designed to self-report and manage hypertension and diarrhea using remote monitoring.
In 2018, the results were published in JCO Clinical Cancer informatics. The authors (J.Liu et al), highlighted feasibility, usability and perceived satisfaction with the use of the digital therapeutic and related healthcare team web portal.
Patients provided positive responses on the use of the eCO app. They indicated feeling more involved in their care and better connected with their healthcare team. Hypertension and diarrhea events reported via the eCO app allowed rapid response and positive overall patient experience.
eCO captured 98,2% of required blood pressure measures (4)
of diarrhea events
limited to grade 1 (4)
*eCO is a digital therapeutic for patients with ovarian cancer that has been designed for investigational purposes. eCO is not commercially available.

